 Osaka Medical and Pharmaceutical University, Fukui Tateami Co., Ltd. and Teijin Limited announced today that they have achieved the primary efficacy endpoint in an ongoing clinical trial of the OFT-G1 (provisional name) patch for cardiovascular surgery. The trial, which began in May 2019 to examine patients ranging from infants to adults suffering from congenital heart diseases, has demonstrated the safety and efficacy of the patch in one-year periods following surgery.
Osaka Medical and Pharmaceutical University, Fukui Tateami Co., Ltd. and Teijin Limited announced today that they have achieved the primary efficacy endpoint in an ongoing clinical trial of the OFT-G1 (provisional name) patch for cardiovascular surgery. The trial, which began in May 2019 to examine patients ranging from infants to adults suffering from congenital heart diseases, has demonstrated the safety and efficacy of the patch in one-year periods following surgery.
The three partners now plan to apply for approval and launch the OFT-G1 patch for cardiovascular surgery as quickly as possible in Japan and then other countries worldwide. Simultaneously, they will continue enhancing the product’s capacity to contribute to the quality of life of patients with cardiovascular diseases.
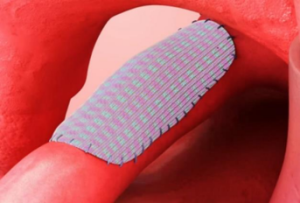
The patch is expected to alleviate physical, mental or financial burdens that patients and their families may experience in connection with corrective surgeries. It features a revolutionary design developed with Osaka Medical and Pharmaceutical University’s expertise in cardiovascular surgery, Fukui Tateami’s pioneering warp-knitting technology and Teijin’s advanced polymer-application technology.
The patch, which consists of a knitted fabric made with biodegradable and non-biodegradable yarns, is coated with a bio-absorbable cross-linked gelatin. Its biotechnological design is expected to enhance the regeneration of natural tissue in patients and adaptability to growing organs in young patients.
Development was supported by a program launched in 2017 by the Japan Agency for Medical Research and Development (AMED) to facilitate collaboration between medical academia and industry. Also, in April 2018, Japan’s Ministry of Health, Labor and Welfare designated the OFT-G1 as a “SAKIGAKE” product qualifying for various incentives to speed up approval for clinical use.
Treatments have been devised to improve the long-term survival of patients who undergo cardiovascular surgery for purposes such as replacing defective heart tissue or enlarging narrowed blood vessels (stenosis). Demands are growing for advanced treatments that are less likely to cause stenosis due to the patient’s somatic (physical) growth in areas where patches have been implanted, thereby eliminating needs for follow-up operations to replace patches that have deteriorated or calcified after long-term implantation.